
Although widely used, fluorescent measurement of ROS is notoriously problematic, especially with regards to measurement of ROS induced by specific and not mitogenic stimuli. In this paper, we demonstrate the use of fluorescent probes and flow cytometry to detect ROS production. Being able to measure ROS production reproducibly and with ease is necessary in order to assess the contribution of various pathways and molecules to this mechanism of host defense. ROS generated during immune activation exerts potent antimicrobial activity primarily through the ability of ROS to damage DNA and proteins, causing death of microorganisms. We provide experimental design consultation for free.The oxidative or respiratory burst is used to describe the rapid consumption of oxygen and generation of reactive oxygen species (ROS) by phagocytes in response to various immune stimuli.Non-NYMC users are charged 50% more for all services.After the initial hour, users are charges on half-hour increments. All services are billed for a minimum of one hour.Several hundred of specific protocols describing applications of cytometry for particular research projects, published in the Current Protocols in Cytometry, are also available to the users of the core laboratory to plan their experiments. Investigators considering flow cytometry service should contact the laboratory for free consultations. The laboratory can also help investigators to explore potential new applications that may be of special value in their research projects. 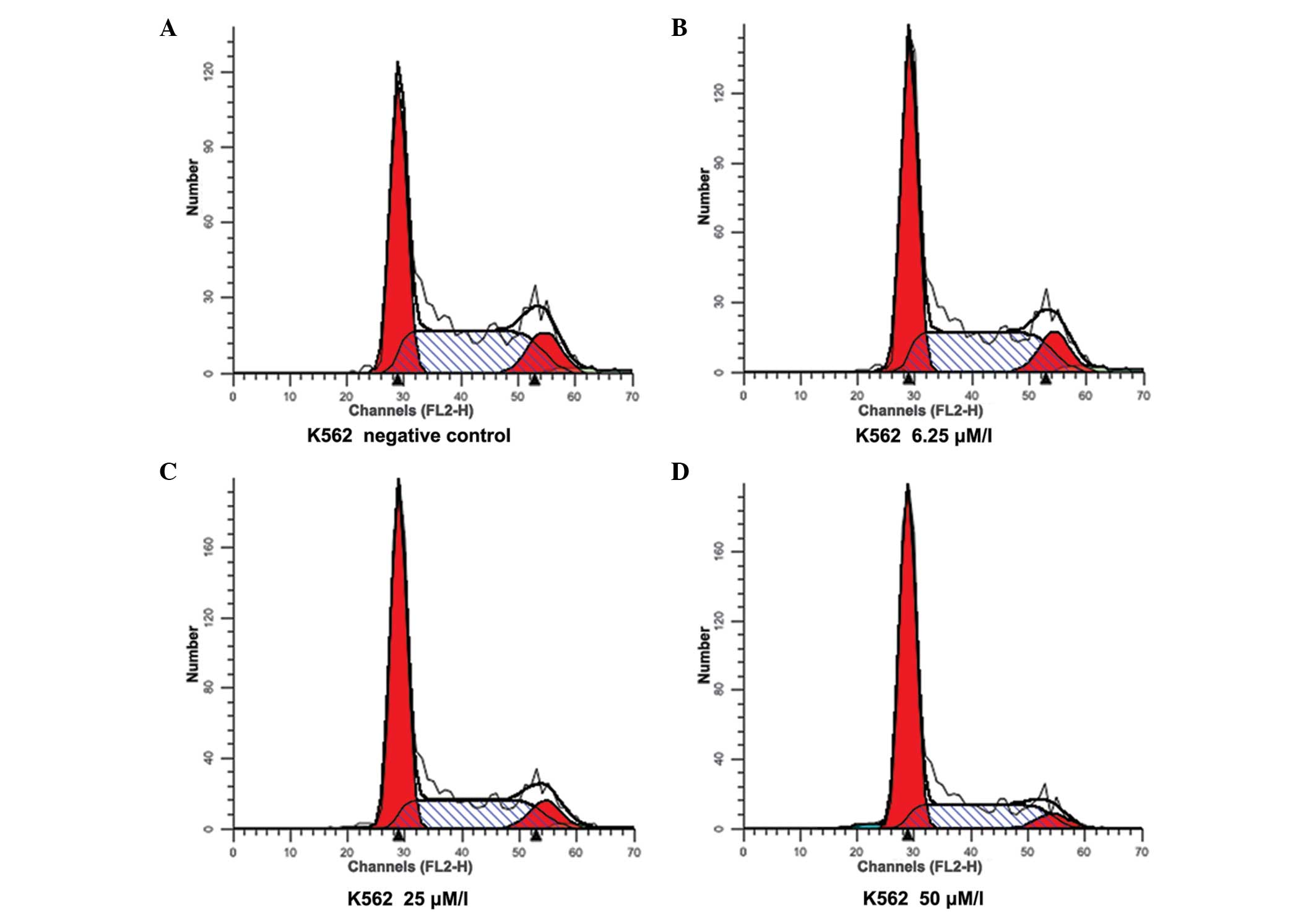 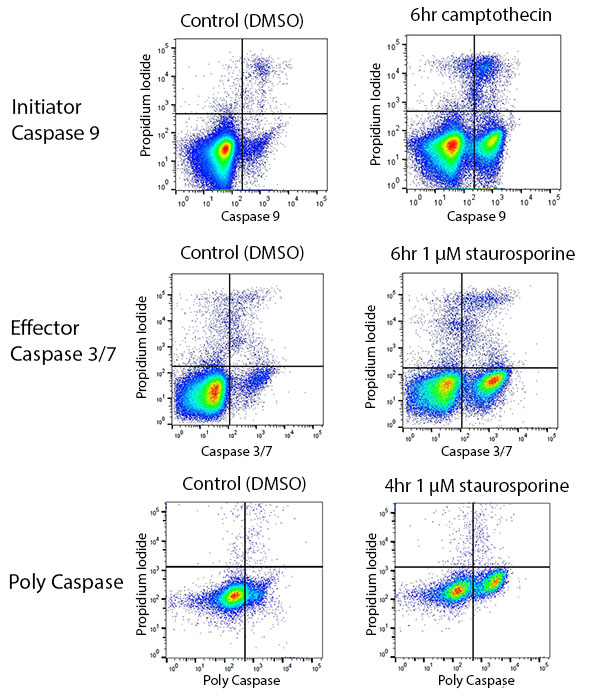
These include project consultation, sample preparation, sample analysis, and data analysis. The laboratory provides high quality comprehensive services on flow cytometry, FACS, and scanning cytometry analyses. Potential applications of scanning cytometry include the measurement of apoptosis, cell cycle progression, DNA damage, senescence, cell viability, and protein translocation within cells.Ĭomputerized flow cytometry data acquisition and analysis systems coupled with most up-to-date software packages (FlowJo, Kaluza, and Multicycle) are available in the laboratory. The instrument operates at a rate up to a hundred cells per second and has the capacity to revisit cells, record cell images, measure live and fixed cells on the slide and create a photomontage of selected cells. The laboratory is also equipped with a CompuCyte four laser (violet, blue, yellow and red) iCys scanning cytometer for four color analysis of fluorescent stained cells on slides and ratios of the various parameters. Also available in the laboratory is a Becton Dickinson FACScan flow cytometer for three-color analysis. The instrument is equipped with four way sorting and the ability to sort single cells into multiwell plates under sterile conditions. The system can acquire two light scatter parameters (forward and right angle) and eleven fluorescence measurements from violet (405 nm) to near infrared (785 nm). The laboratory is equipped with a Beckman Coulter MoFlo XDP high-speed flow cytometer/sorter equipped with three laser light sources for simultaneous excitation in the UV, blue and red. research facility in the Department of Pathology, Microbiology, and Immunology. The Flow Cytometry Core Laboratory of New York Medical College is located in the Brander Cancer Research Institute, a 4,300 sq. New applications of flow cytometry are still being developed constantly. Research fields such as immunophenotyping, cell cycle, apoptosis, cell signaling, and cell sorting/isolation are well established areas of application of these techniques. These powerful techniques have broad research and clinical applications. Fluorescence-activated cell sorting (FACS) enables researchers to sort live cells rapidly according to their physical characteristics or cell surface markers so that subpopulations of cells can be studied separately. Flow cytometry allows rapid analysis, and classification of cells or other small particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
